Blood Pressure Drug Recalled After Cancer-Causing Chemicals Detected: What You Need to Know
Blood Pressure Drug Recalled After Cancer-Causing Chemicals Detected: What You Need to Know

A widely used blood pressure medication has recently raised serious concerns after being recalled due to the detection of potentially cancer-causing substances. This development has left many patients questioning the safety of their medications and wondering what steps they should take next.
The recall involves drugs commonly prescribed to control high blood pressure and reduce the risk of heart disease. In several cases, testing revealed the presence of chemical impurities known as nitrosamines, which are classified as probable carcinogens. These substances, including NDMA and NDEA, have been linked to an increased risk of cancer when exposure occurs over long periods.
Health authorities, including the U.S. Food and Drug Administration, have emphasized that the issue is not with the medication itself, but with contamination during the manufacturing process. In many instances, changes in production methods or poor quality control allowed these harmful compounds to form unintentionally.
Millions of people rely on medications like valsartan, losartan, and other similar drugs to manage their blood pressure. These medications are essential for preventing serious conditions such as heart attacks, strokes, and heart failure. However, the discovery of contaminants has triggered widespread recalls across multiple countries and manufacturers.
Despite the alarming headlines, experts urge patients not to panic. The actual risk depends on factors such as dosage, duration of use, and individual health conditions. In many cases, the increased cancer risk is considered relatively low, especially for short-term exposure. However, long-term use of contaminated medication could potentially pose a greater concern.
One of the most important warnings from health officials is this: do not stop taking your medication suddenly without medical advice. Abruptly discontinuing blood pressure medication can cause serious complications, including spikes in blood pressure that may lead to heart attacks or strokes. Instead, patients are advised to consult their doctor or pharmacist to determine whether their specific medication is affected and to receive a safe replacement if necessary.
This situation also highlights a deeper issue within the global pharmaceutical supply chain. Many generic drugs are manufactured using ingredients sourced from overseas facilities. When quality control fails at any point in the process, the consequences can affect millions of people worldwide.
Ultimately, this recall serves as a reminder that even widely trusted medications require strict oversight. Patients should stay informed, check official recall lists, and maintain open communication with healthcare providers.
Your health decisions matter — and sometimes, the smallest detail can make the biggest difference.
News in the same category


Many people believe that storing leftovers in the fridge automatically makes them safe to eat the next day

Doctors reveal that eating avocado causes... see more
5 Alarming Stroke Warning Signs to Watch for in Young People

Is He Pulling Away? The Real Reason Your Partner Turns Their Back While Sleeping

If Your Cat Sleeps Next to You, It Might Be Trying to Tell You Somethin
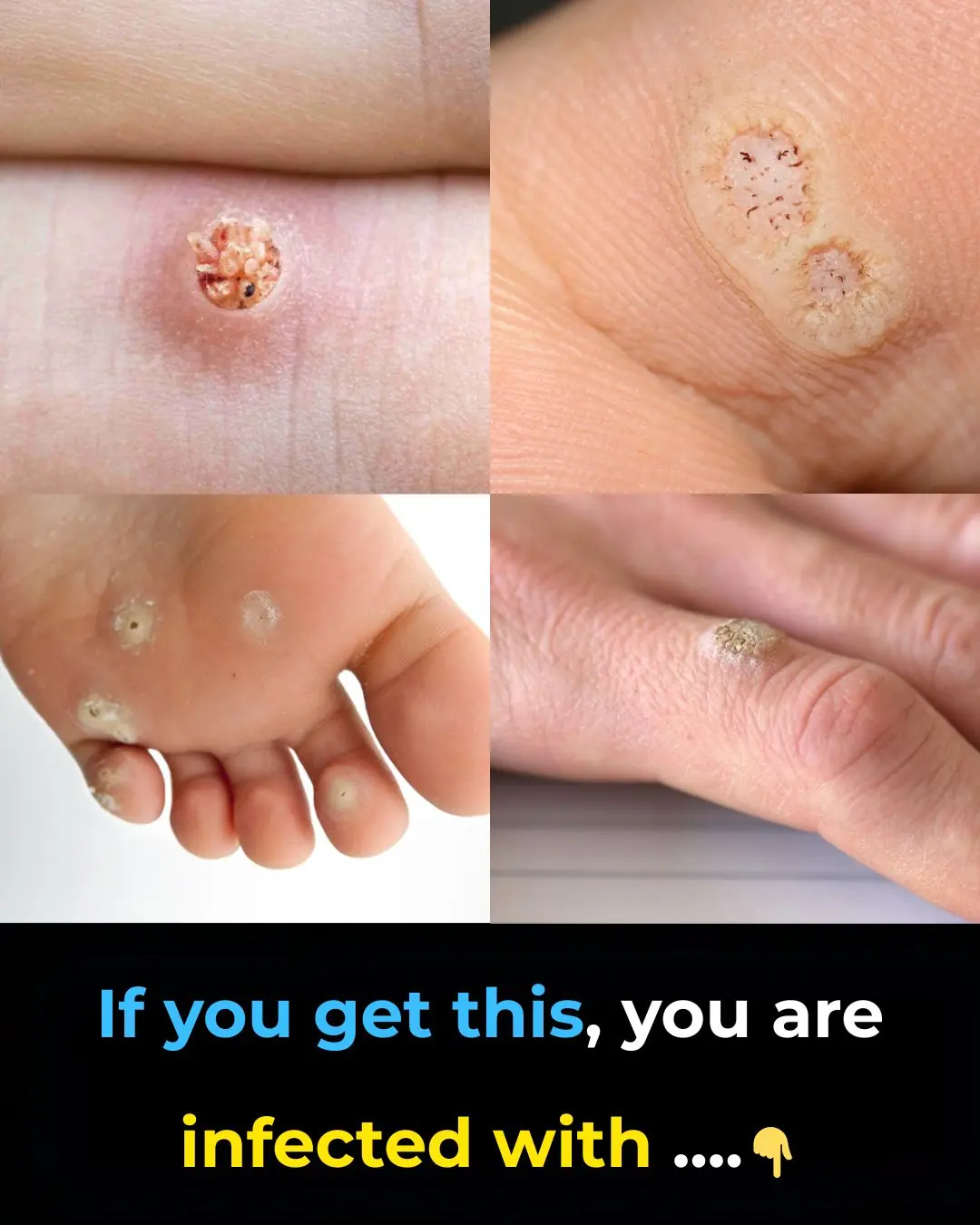
6 Types of Warts: Signs, Symptoms, Home Remedies, and Treatment
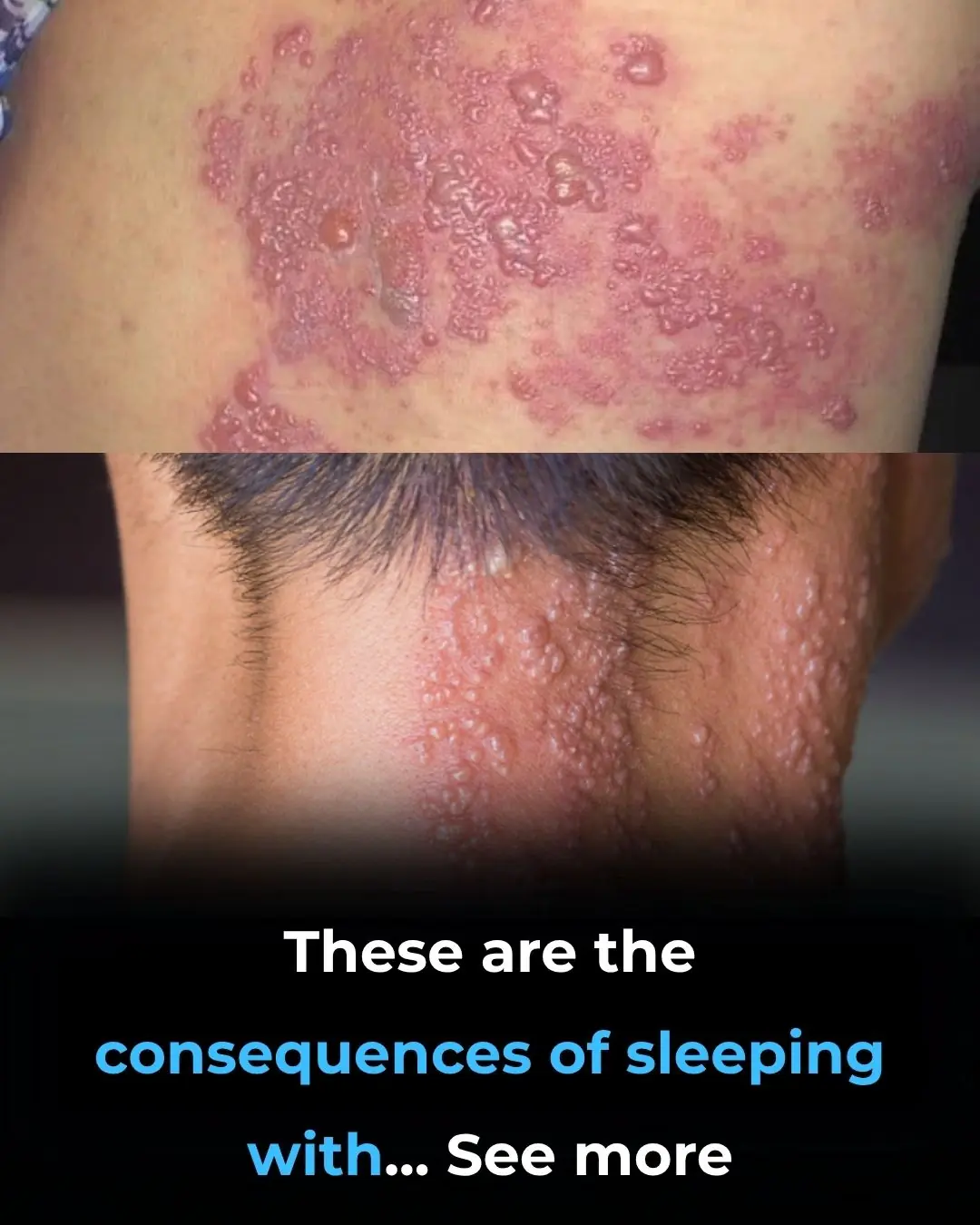
These are the Consequences of Sleeping with the Fan On

This Simple Night Routine May Help Protect Your Heart (And Most People Ignore It)

Cancer Often Shows No Pain at First – But These 8 Bathroom Warning Signs Should Never Be Ignored

8 of the Best Anti-Cancer Foods: It’s Time to Start Adding Them to Your Diet

Why Kidney Failure Is Striking the Young—And How to Stop It

Coughing Caused by Acid Reflux: Hidden Signs, Causes, and Effective Solutions
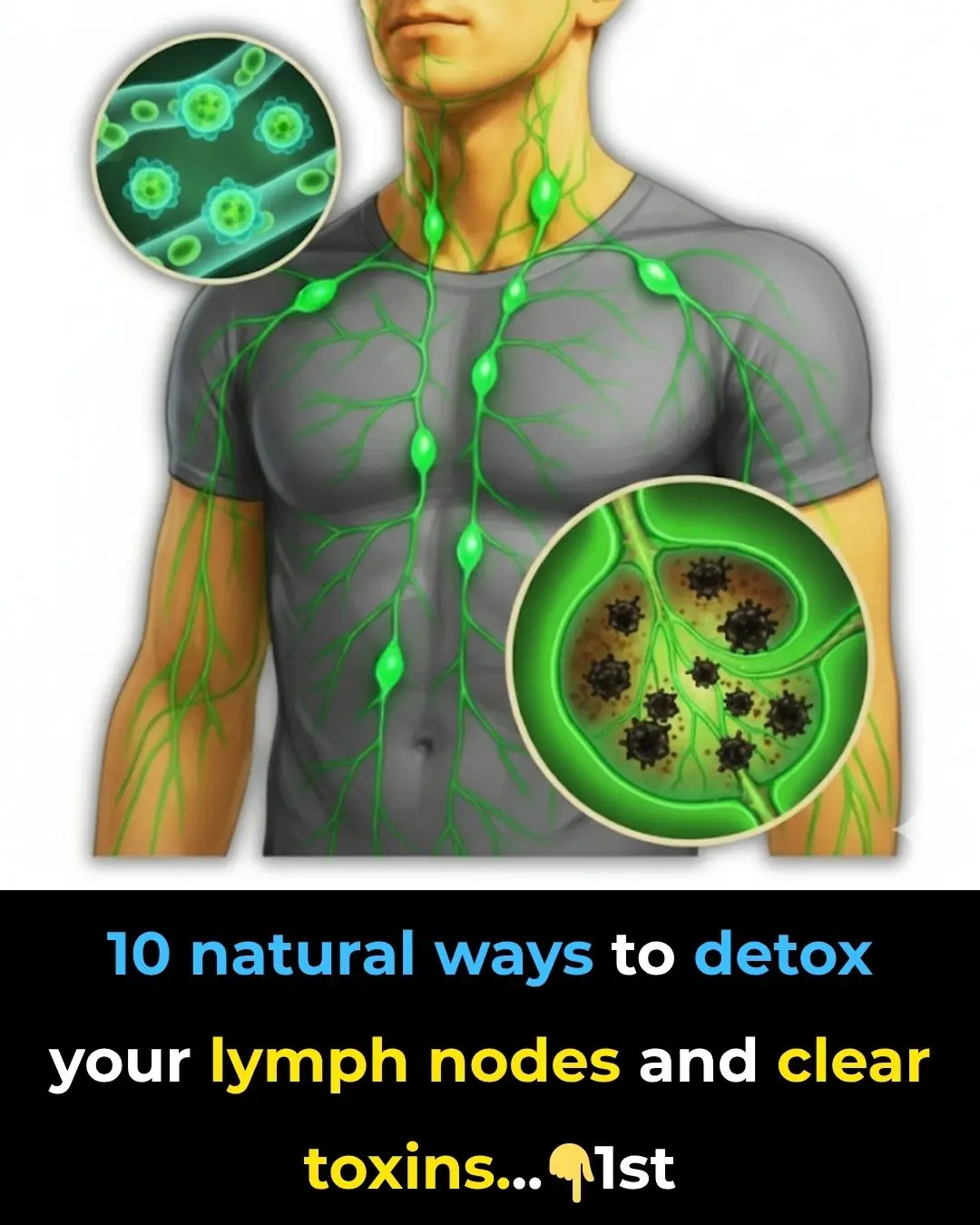
Deep Diaphragmatic Breathing

Tonsil Stones: Causes, Symptoms, and Solutions

Not Just Forgetfulness… Your Brain May Be Warning You

Summary: New Scientific Findings on the Causes of Autism
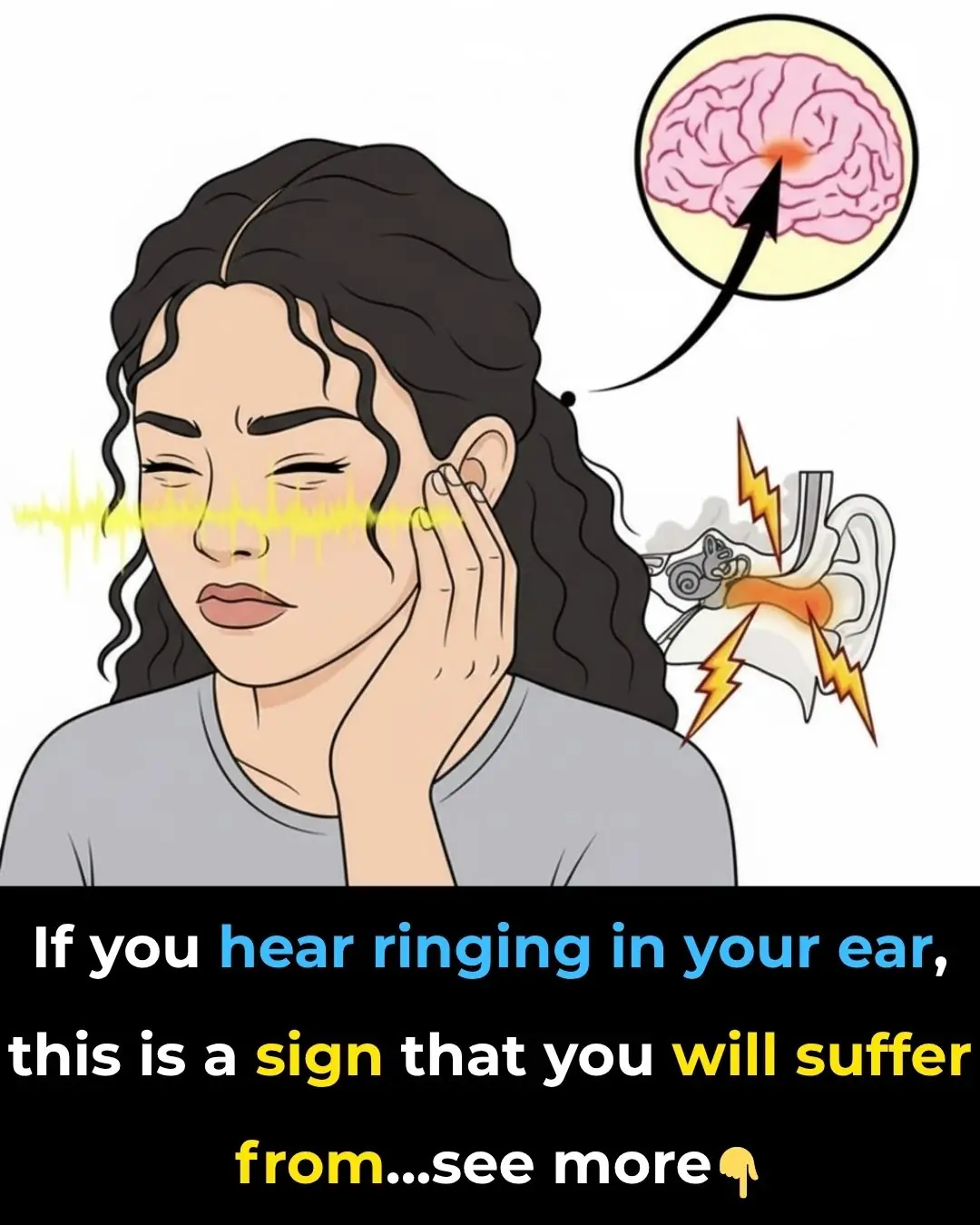
Ringing in Your Ear: Understanding Tinnitus

Say Goodbye to Joint and Foot Pain with a Relaxing Rosemary Bath
News Post

The Viral Video That Changed Everything. A Dog, a Family, and the Truth That Came Out

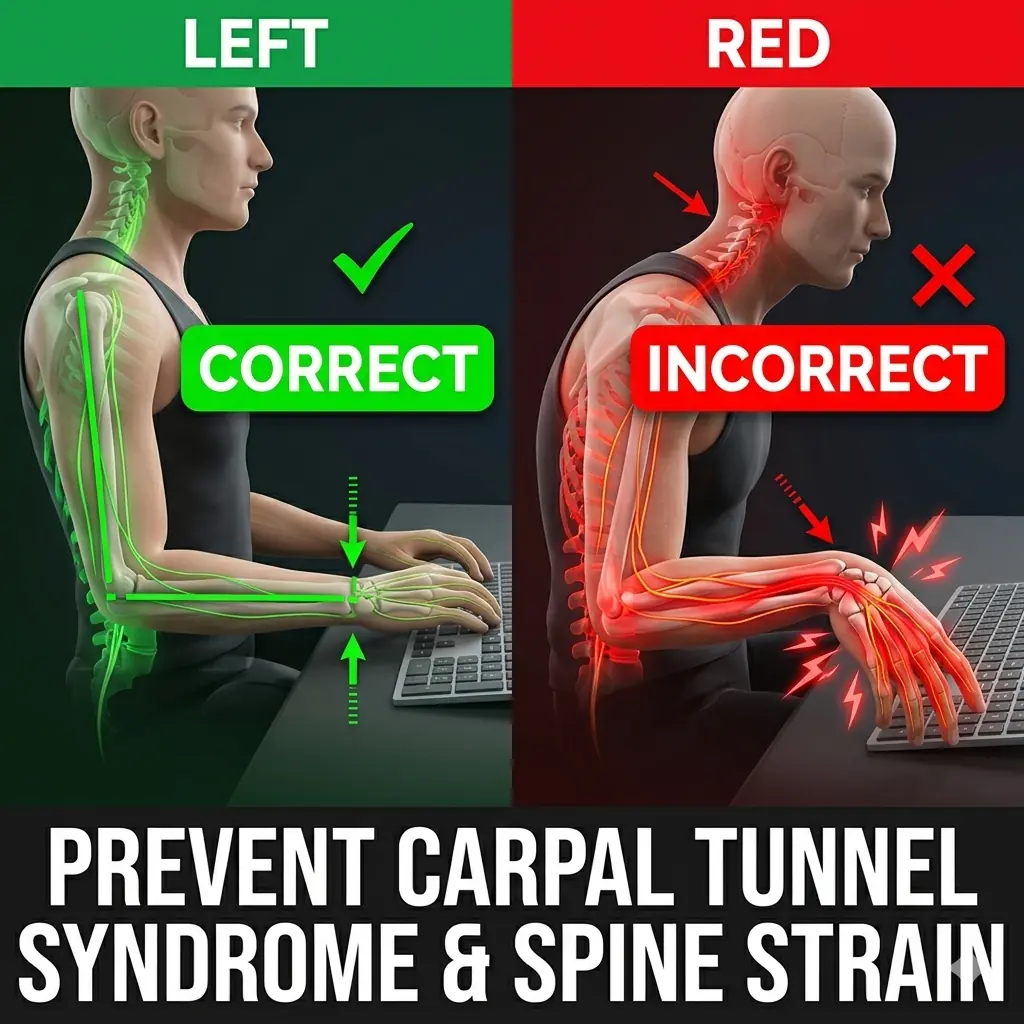
Correct vs Wrong Sitting Posture: How Poor Wrist Position Can Damage Your Nerves

The Arsenal of Democracy in Crisis: Why the US Needs China to Rebuild Its Weapons Stockpiles

US-Iran Geopolitical Stalemate: Why Neither Side Can Sustain a Long-Term War

Cop Assaults Black Navy SEAL At Grocery Store — Security Camera Exposes Him

The Boy Who Saw the Truth

An Elderly Woman Tried to Pawn Her Wedding Ring to Save Her Sick Son — But What the Shop Assistant Did Left Everyone Stunned

She Was Judged by Her Clothes — Until the Truth Left the Restaurant in Shock

The Police Dog Wouldn’t Stop Barking at a Homeless Man. Seconds Later, Everyone Understood Why

In the courtroom, a woman rose from the defendant’s bench and stated in a calm voice that her dog would prove her innocence.

The day when, under a lead-colored sky, a dog began to look passersby in the eyes with such despair that even the busiest people stopped.

Many people believe that storing leftovers in the fridge automatically makes them safe to eat the next day

The 15th Floor Nightmare: When Man’s Best Friend Isn’t Human

The Mistress Poured Water on a “SERVANT” and Said I’d Never Be More Than Dirt—She Had NO IDEA Who I Really Was

The Bride SHOVED a 5-Year-Old to the Floor and SHREDDED His Drawing — Then Her Face Turned WHITE

The Groomsman Mocked a Quiet, Overweight Chef During the Cake Cutting — Then His Face Turned WHITE

Abandoned by Her Family, Maria Braved the Dangerous River — Unaware That Something Far More Terrifying Awaited on the Other Sidev

The First 3 Colors You See Reveal Your Personality (This Test Is Surprisingly Accurate)
