
Tips 16/03/2026 21:55
Scientific decoding: Why are lollipops actually "edible glass"?
News in the same category

Tips 16/03/2026 17:12

Your Choice May Reveal Something Interesting About Your Personality
Tips 16/03/2026 16:23

A SCIENTIFIC GUIDE TO NATURALLY INCREASING MALE VITALITY
Tips 16/03/2026 15:37

The Secret Purpose of the Hole in a Safety Pin: Why 98% of People Get It Wrong
Tips 16/03/2026 14:34

Can You Eat Yogurt Past Its Expiry Date
Tips 16/03/2026 10:44

5 Types of Women Men Never Truly Forget
Tips 16/03/2026 10:41

Beyond the "Hero" Mask: The Silent Struggle of Men’s Performance
Tips 16/03/2026 09:37
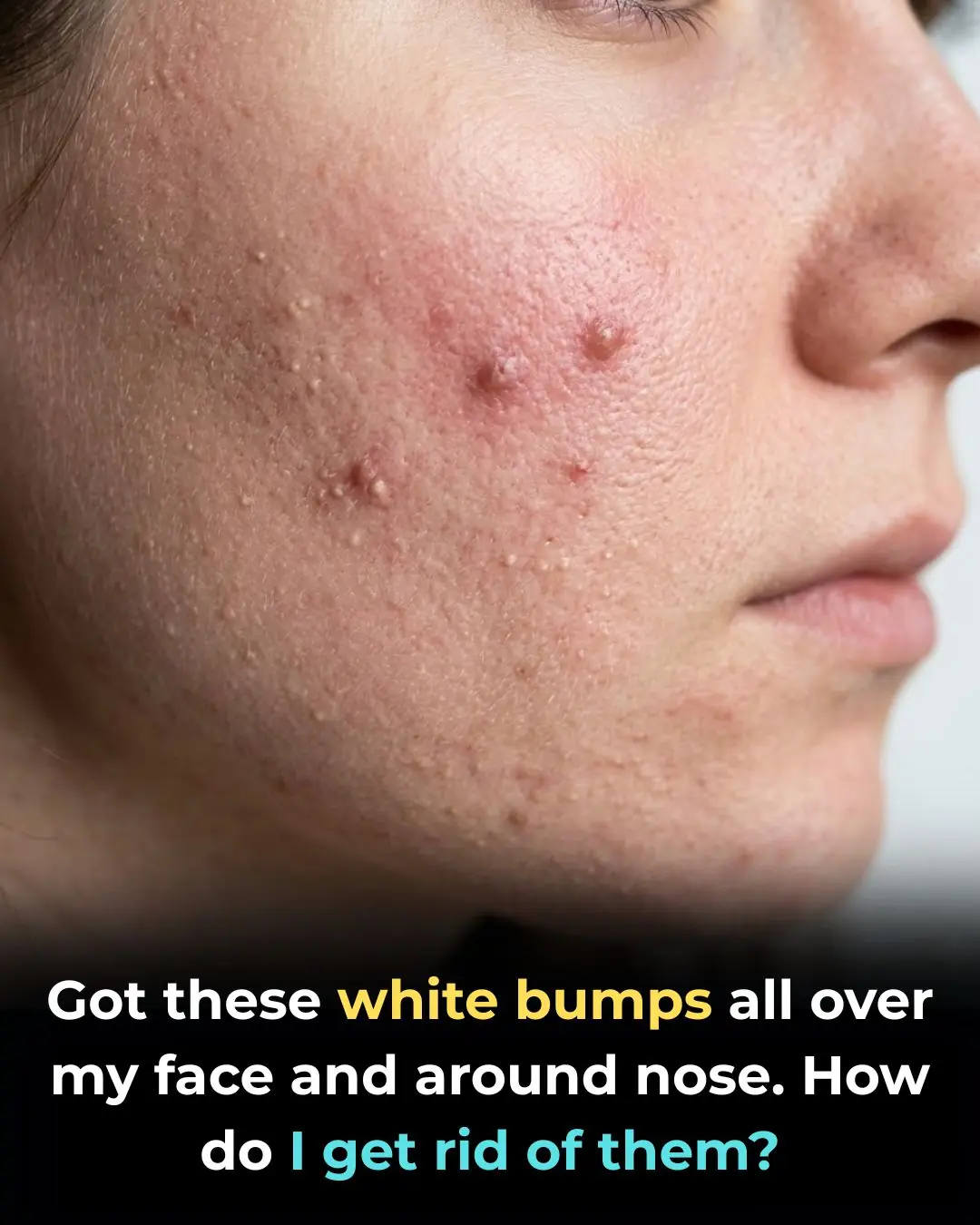
I truly am bothered by these!
Tips 14/03/2026 08:45

The Silent Echo in the Hallway: A Story of Love, Neglect, and the Way Home
Tips 13/03/2026 16:52
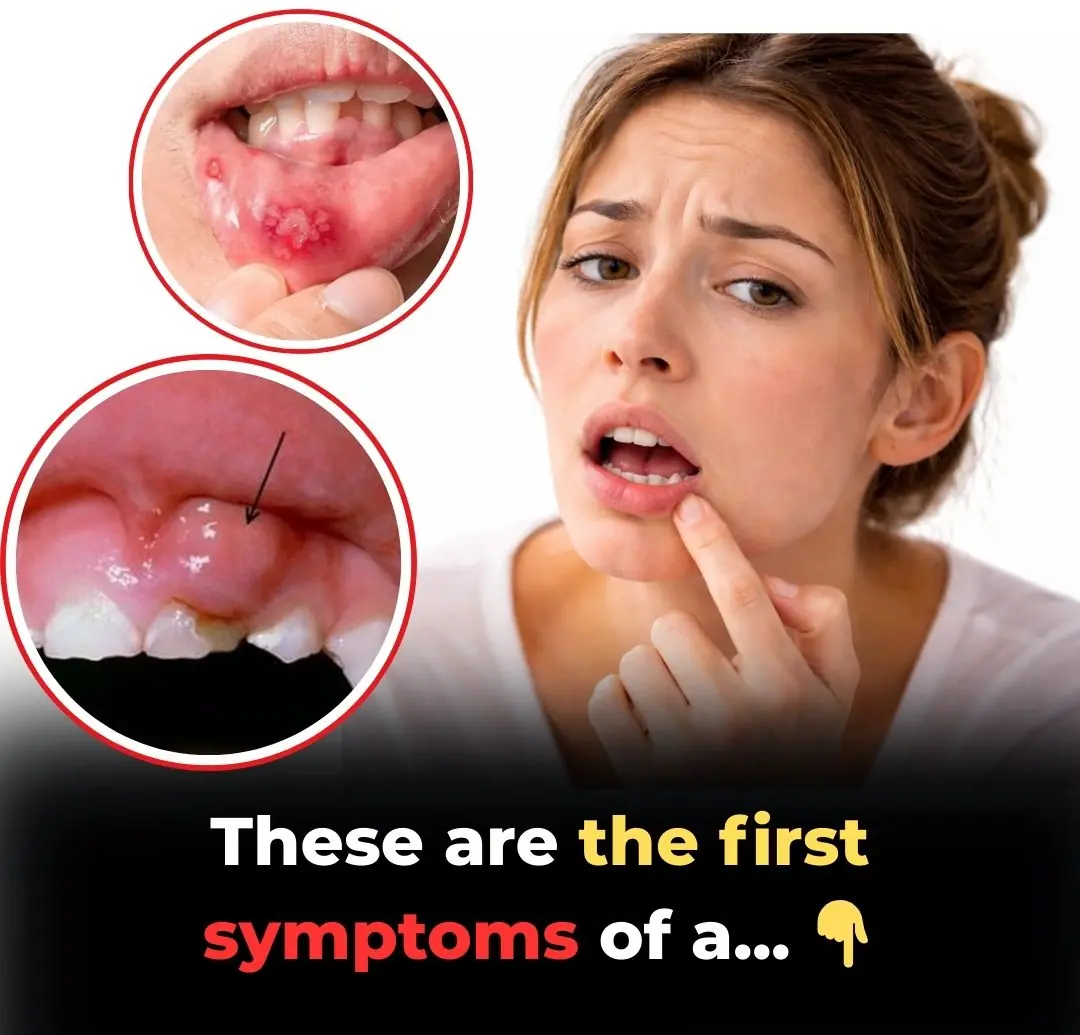
Is Your Mouth Trying to Tell You Something? 5 Hidden Warning Signs of Oral Cancer
Tips 13/03/2026 11:46

Your choice reveals your personality...
Tips 13/03/2026 08:18

What Left-Handed People Have That Right-Handers Don’t
Tips 11/03/2026 22:03

Never Ignore Excessing Licking From a Dog
Tips 11/03/2026 13:40

10 Bizarre Home Remedies
Tips 10/03/2026 20:53

When making pickled garlic, you need to add this extra step — the garlic stays white and crunchy,
Tips 10/03/2026 14:01

Put an Empty Plastic Bottle in the Washing Machine?
Tips 10/03/2026 13:58

Soak Lemon Peels in Vinegar to Create a Powerful Natural Solution with Surprisingly Useful Benefits
Tips 08/03/2026 22:05

What Happens to Your Body When You Eat Cottage Cheese Regularly
Tips 07/03/2026 22:42

7 Foods To Help You Live a Longer, Healthier Life
Tips 07/03/2026 22:38
News Post

Is He a Foe… or an Ally?
Life stories 16/03/2026 22:54

I Was Mourning My Twins at Their Grave When a Boy Said, “Mom… Those Girls Are in My Class
Life stories 16/03/2026 22:36

FOR SEVEN YEARS YOU ATE DINNER IN THE DARK… UNTIL A JANITOR’S TODDLER SAT IN YOUR SEAT AND REWROTE YOUR LIFE
Life stories 16/03/2026 22:27
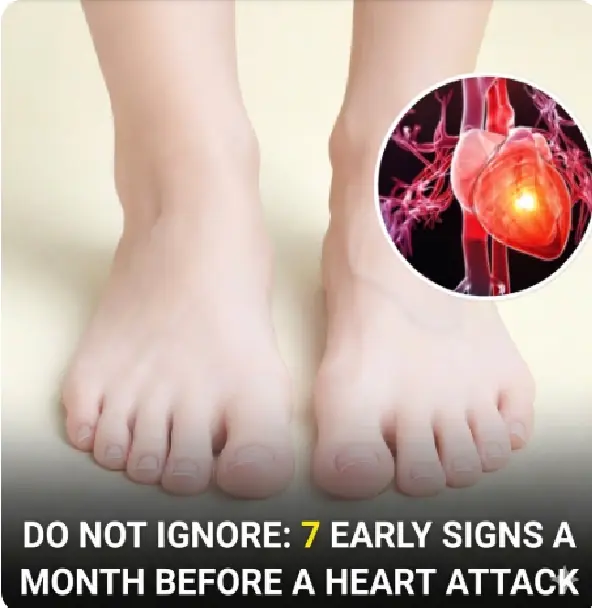
7 Warning Signs of a Heart Attack One Month Before It Happens: Do Not Ignore These
Health 16/03/2026 22:27

SHE WAS THROWN INTO THE SNOW FOR BEING “INFERTILE”… THEN A WIDOWED CEO WHISPERED, “COME WITH ME.”
Life stories 16/03/2026 22:24

BILLIONAIRE WAS TAKING HIS FIANCÉE HOME UNTIL HE SAW HIS EX CROSSING THE CROSSWALK WITH TWINS
Life stories 16/03/2026 22:21

Wife Gave Birth by C-Section — Husband Divorced Her, Mistress Wore Wife’s Wedding Gown Unaware She
Life stories 16/03/2026 22:14

At the airport parking lot, I found my son sleeping in his car with his twins
Beatuty Tips 16/03/2026 21:26

Foamy Urine: Causes, Health Risks, and When to See a Doctor
Health 16/03/2026 21:26

To pay for my father’s cancer treatment, I married a rich old man. On our first night, he removed his mask and said, ‘Now you’ll see who you really married.’
Beatuty Tips 16/03/2026 21:22

He Erased His Wife from the Billionaire Gala — Until the Entire Room Rose When She Walked In
Beatuty Tips 16/03/2026 21:18

YOU COME HOME EARLY FOR LUNCH—AND CATCH YOUR MAID RAISING YOUR QUADRUPLETS LIKE YOU NEVER DID
Beatuty Tips 16/03/2026 21:15

HE DUMPED ICE WATER ON “THE POOR WOMAN”… THEN HE HEARD HER NAME AND THE WHOLE FLOOR WENT DEAD SILENT
Beatuty Tips 16/03/2026 21:10

The mafia boss spent the day with his mistress, silencing his wife’s calls and assuming nothing was wrong. He never imagined that when he finally checked his phone, it would be the last message he’d ever receive from her—a final, irreversible goodby
Beatuty Tips 16/03/2026 21:06

On my wedding night, I hid under the bed to play a prank on my husband, but someone else entered the room and put their phone on speaker. What I heard next made my blood run cold…
Beatuty Tips 16/03/2026 21:03

Are You the One?
Life stories 16/03/2026 21:03

The Picture in His Wallet
Life stories 16/03/2026 20:07

The Runway Miracle Kid
Life stories 16/03/2026 18:37

The Father Who Returned After Fifteen Years—Only to Find His Daughter Living as a Servant in the Mansion He Bought for Her
Life stories 16/03/2026 18:09