
Blood Pressure Drug Pulled From Shelves After Carcinogen Detected
Doctors Warn: A Common Blood Pressure Drug Pulled From Shelves — What They Found Inside Is Raising Concern
Millions of people rely on blood pressure medication every day to keep their health under control. For many, it’s part of a routine — something taken without a second thought.
But recently, that routine has been shaken.
👉 A widely used blood pressure drug has been pulled from shelves after a potential cancer-causing substance was discovered.
And now, both doctors and patients are asking the same question:
How serious is this — and what should you do if you’ve been taking it?
The Drug At The Center Of Concern
The medication involved is prazosin hydrochloride, a commonly prescribed drug used to treat high blood pressure.
Doctors often prescribe it because it works by:
- Relaxing blood vessels
- Improving blood flow
- Reducing strain on the heart
In some cases, it’s also used to help patients with PTSD manage nightmares and sleep disturbances.
That’s why this recall has raised so many eyebrows — this isn’t a rare or obscure medication.
It’s something hundreds of thousands of people use regularly.
What Triggered The Recall?
The issue began when testing revealed something unexpected.
👉 Certain batches of the drug were found to contain nitrosamine impurities — chemicals that are considered potential carcinogens (cancer-causing substances).
Even more concerning:
👉 The levels detected were higher than what safety guidelines allow, which led to immediate action.
As a result, manufacturers voluntarily recalled the medication nationwide, removing large quantities from circulation.
How Big Is The Recall?
This wasn’t a small issue.
- Over 580,000 bottles of the medication were affected
- Multiple dosage levels were included (1mg, 2mg, 5mg)
- Distribution covered a wide area across the U.S.
In short:
👉 This recall impacted a massive number of patients and pharmacies.
What Are Nitrosamines — And Why Are They Dangerous?
Nitrosamines are a group of chemicals that can form during:
- Drug manufacturing
- Storage
- Chemical reactions over time
While small amounts may be harmless, long-term exposure to higher levels has been linked to an increased risk of cancer.
This is why regulatory agencies like the FDA take them very seriously.
In this case, the impurity detected exceeded acceptable safety limits — which is what triggered the recall.
Should You Be Worried?
Here’s where things get important.
The recall was classified as a Class II risk, meaning:
👉 It may cause temporary or reversible health effects
👉 The chance of serious harm is considered low, but not zero
So while the word “cancer” sounds alarming, experts emphasize that:
- Risk depends on dose and duration of exposure
- Not everyone who took the medication is in danger
Still, it’s not something to ignore.
What Doctors Are Urging Patients To Do
One of the biggest mistakes people make after hearing news like this is panic.
👉 Doctors strongly advise: DO NOT stop taking your medication suddenly.
Stopping blood pressure medication abruptly can:
- Cause dangerous spikes in blood pressure
- Increase the risk of stroke or heart attack
Instead, experts recommend:
✔ Contact your doctor or pharmacist
✔ Check if your medication batch is affected
✔ Ask for a safe alternative if needed
Why This Keeps Happening
This isn’t the first time medications have been recalled due to carcinogen concerns.
Over the past few years, similar issues have been found in:
- Blood pressure drugs
- Heartburn medications
- Other widely used prescriptions
These cases often trace back to:
- Manufacturing processes
- Chemical instability over time
👉 It highlights a bigger issue: even regulated medications are not completely risk-free.
The Bigger Lesson Most People Miss
Many people believe that if a drug is approved, it’s 100% safe.
But the reality is more complex.
Medications are:
- Tested before approval
- Continuously monitored after release
Sometimes, problems only appear after years of widespread use.
That’s why recalls like this happen — not because the system failed entirely, but because it’s still learning and adjusting.
The Bottom Line
So what should you take away from all this?
👉 Yes, a common blood pressure drug was recalled due to a potential cancer risk
👉 The issue involves contamination — not the intended effect of the drug
👉 The overall risk is considered moderate to low, but still important
And most importantly:
👉 Do not panic — but do stay informed.
Final Thought
Sometimes the biggest health risks aren’t obvious symptoms — they’re the things we trust without question.
And moments like this are a reminder:
👉 Always pay attention to what you’re putting into your body
👉 Stay updated on medical news
👉 And never ignore changes that could affect your health
News in the same category


US Deploys B-52 Bombers Over Iran as Military Shifts to 'Dynamic Targets'
The Rising Toll: How Extreme Heat is Redefining Global Health and Work
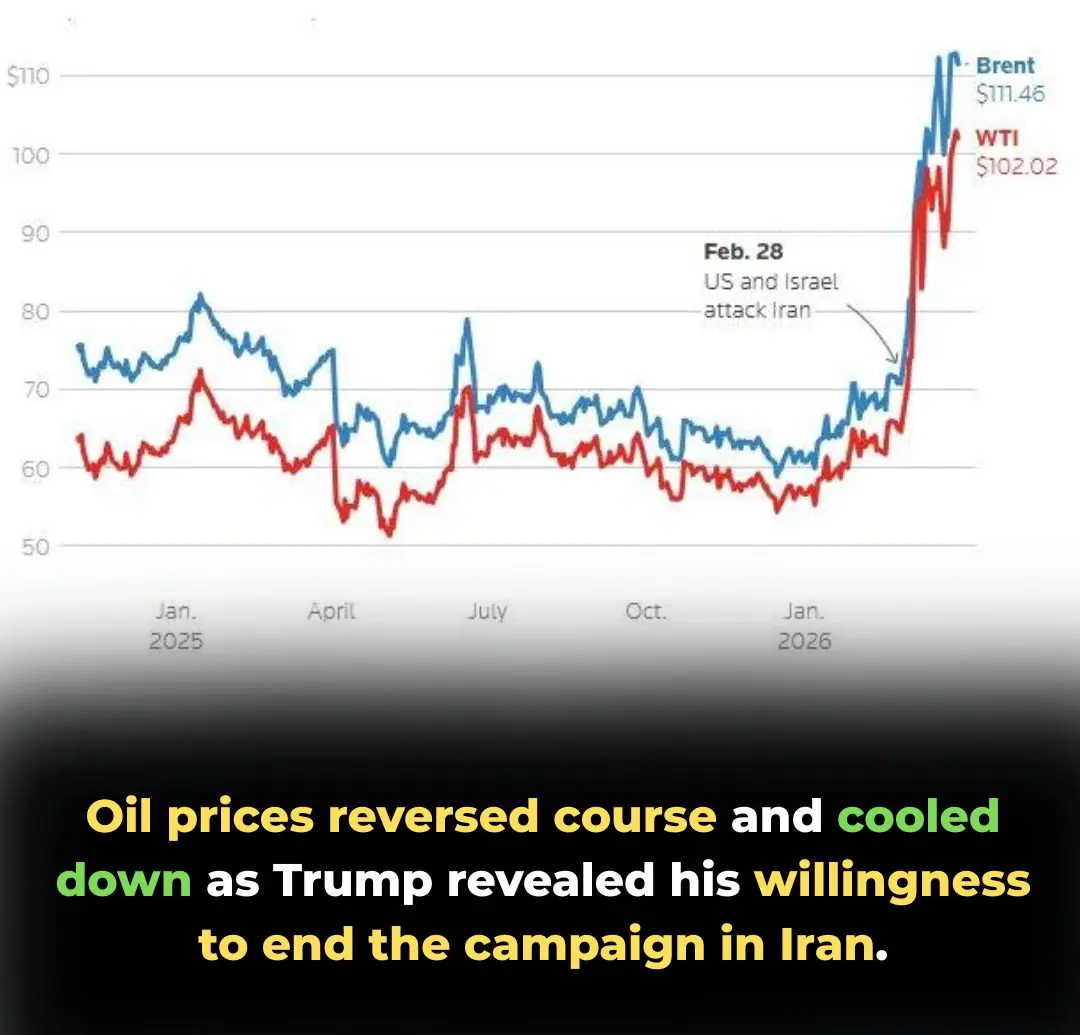
Global Oil Prices Pivot: Markets Cool as Trump Signals Potential End to Iran Campaign

Budget airlines built on cheap fares now face a painful reality: Fuel is getting expensive

China suppliers warn of higher prices for Americans due to Strait of Hormuz closure

Iran "Waiting" for U.S. Ground Forces: Tehran Warns of Lethal Response to Troop Deployment
Oil Markets React to U.S. President’s War Comments — But Traders Remain Cautious

Gold firms on dip-buying; dimming Fed rate-cut hopes cap rise

Oil prices rise with Brent heading for record monthly surge as Iran war enters fifth week

Why This Everyday Habit Is Getting So Much Attention Online

Why Gold’s Role as a Safe Haven is Surgeing in a Fragmented Global Economy: Insights from Swiss Bankers
There are 5 types of feet: each indicating a particular personality type go to the first comment ...👇

WHICH CAR, IF PUT IN REVERSE, WILL SOLVE THIS PROBLEM?

Can You Spot the Hidden Mistake in This Hospital Picture?

Pick The Odd One Out To See How ‘Difficult’ You Really Are...

Oil, gas exploration is back: Energy giants hunt to replenish reserves

Gold demand improves in India as prices ease; China sees softer buying
News Post

I came to his villa for one last chance to save our marriage—not to hear him sneer
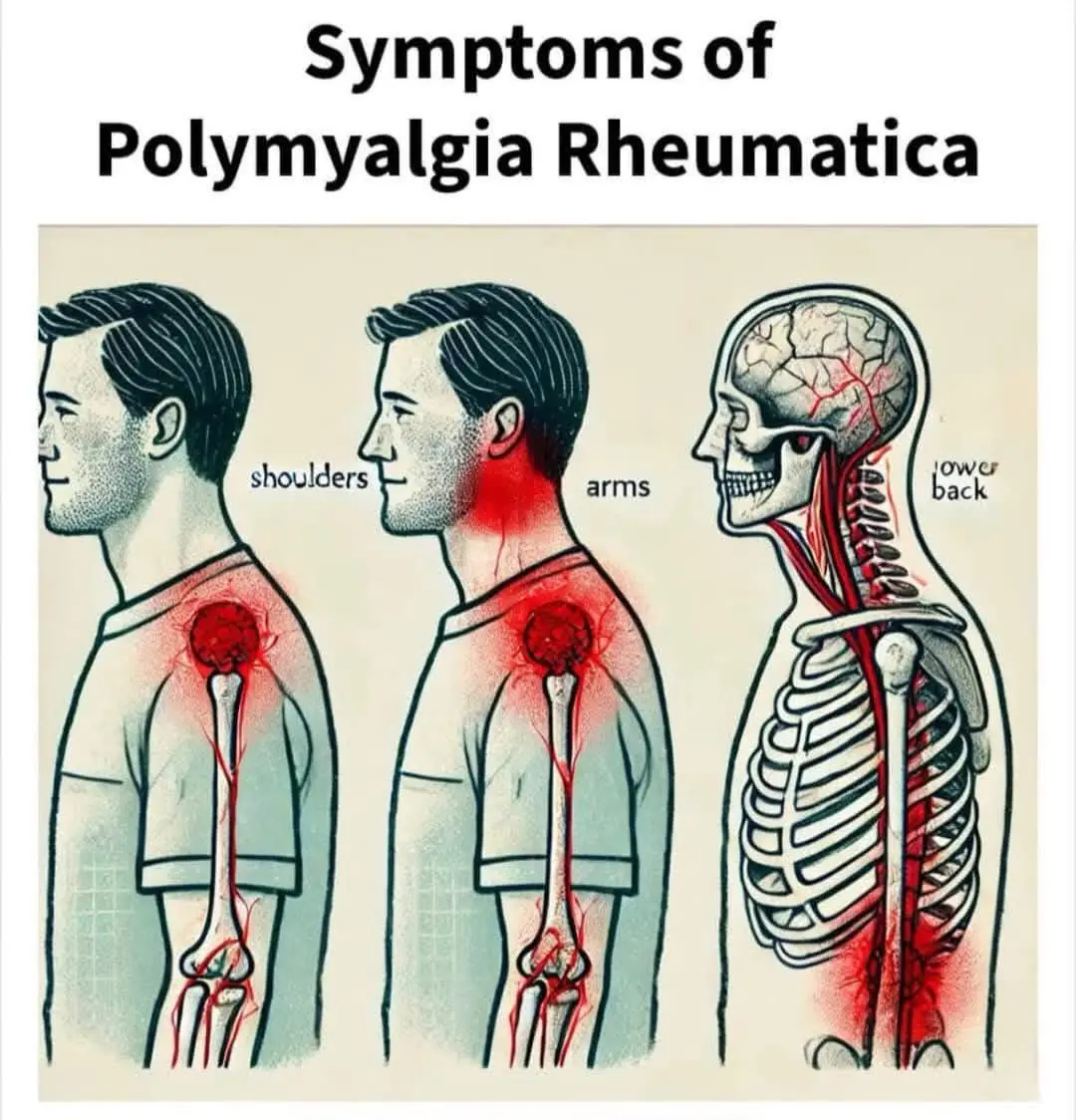
Polymyalgia Rheumatica: Key Symptoms and Indicators

The Surprising Psychology Behind Crossed Legs Habit in Women

Why Dogs May Sniff Your Genital Area: Surprising Truth

The Truth Behind the $400 Million American Bridge That’s Left People Stunned After ‘Disappearing Into the Water’

Reconnecting Hearts: Why Emotional Intimacy is the Secret to Lasting Love

Iran threatens Nvidia, Apple and other tech giants with attack

Doctors Reveal What Happens When You Eat Avocado Regularly — The Truth Might Surprise You

Europe stocks rebound strongly as Trump says Iran war will end in weeks

The Hidden Risks of Falling in Love After 60 – What Experts Rarely Talk About
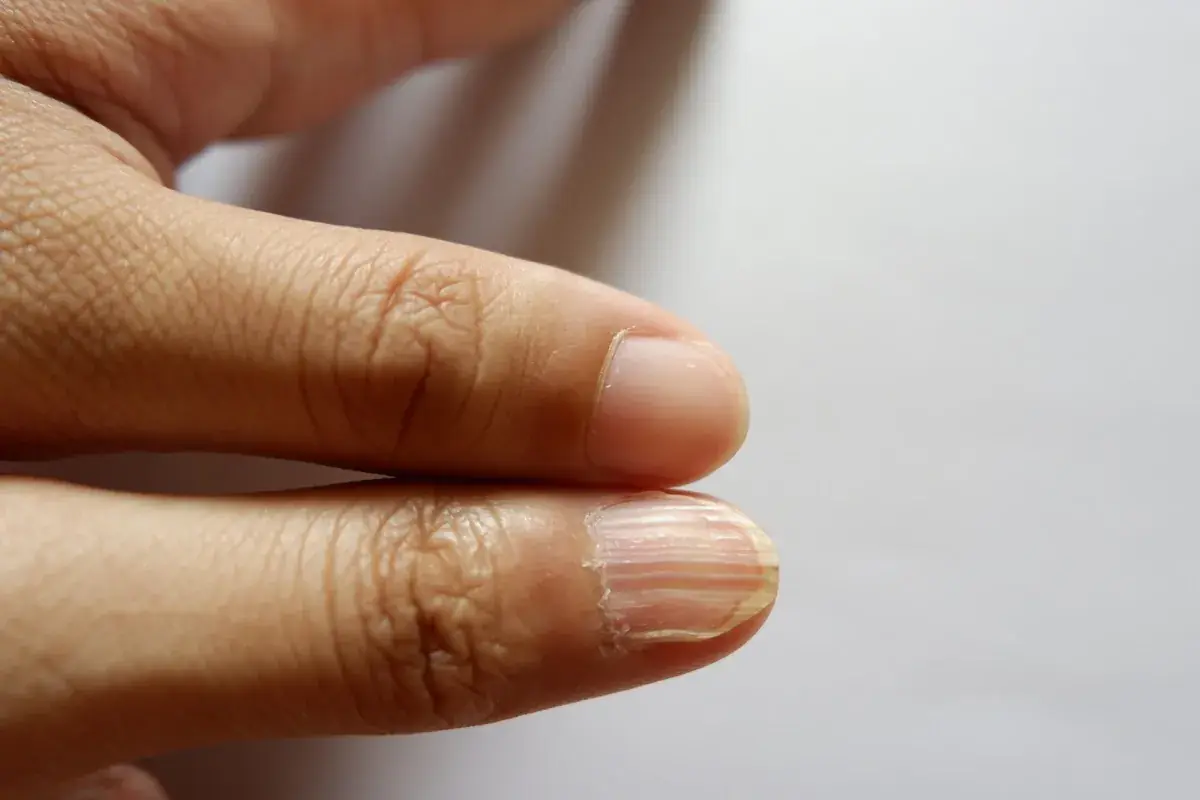
The Hidden Health Clues You’re Missing Every Time You Look at Your Nails

THE FORGOTTEN LIST

How to Solve the Red Bucket and Gas Tank Math Puzzle

Husbands With These 2 Bad Habits May Put Their Wives at Higher Risk of Breast Cancer – Stop Them Now!

The Golden Fruit: Health Benefits and Optimal Ways to Enjoy Oranges

If you enter this room, which chair would you sit in?

US Deploys B-52 Bombers Over Iran as Military Shifts to 'Dynamic Targets'
The Rising Toll: How Extreme Heat is Redefining Global Health and Work
